更新时间:2022-01-28
Plasmocin® is a broad-spectrum mycoplasma and related cell wall-less bacteria removal reagent. It is effective against all common mycoplasma strains, both extracellular and intracellular.
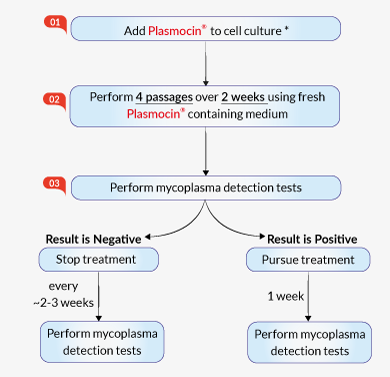
Cell culture treatment using Plasmocin®
 INVIVOGEN ALSO OFFERS
INVIVOGEN ALSO OFFERS• MycoStrip™ — Genomic detection strips
• PlasmoTest™ — Colorimetric cell-based assay
Plasmocin® is a broad-spectrum mycoplasma and related cell wall-less bacteria removal reagent. It is effective against all common mycoplasma strains, both extracellular and intracellular.
For maximum efficiency, Plasmocin® contains a formulation of two antibiotics: the first one blocks protein synthesis, and the second one stops DNA replication. Another component, which is actively transported into mammalian cells, ensures that following treatment, cell cultures do not become re-infected. Thus, Plasmocin® is more effective than other reagents on the market in eradicating mycoplasma and preventing resistant strain generation [1]. Many cell lines infected by mycoplasmas have been successfully treated with Plasmocin® [2], including primary cancer cell lines [3], virus-producing cells [4], and induced pluripotent stem cells [5], without inducing any permanent alterations.
This mycoplasma removal reagent is available in two formats:
Plasmocin® prophylactic is a product at an optimal concentration that can be used as a routine addition in liquid media to prevent mycoplasma and more generally bacterial contamination in mammalian cell cultures.
One 1 ml vial is sufficient for 500 ml of culture.
Plasmocin® treatment is intended for mycoplasma elimination within 2 weeks. It does not affect cell stemness, pluripotency, nor viability [6].
One 1 ml vial is sufficient to treat 660 ml to 2 liters of culture.
References:
1. Molla Kazemiha V. et al. 2011. Efficiency of Plasmocin on various mammalian cell lines infected by mollicutes in comparison with commonly used antibiotics in cell cultures: a local experience. Cytotechnology. Dec; 63(6):609-20.
2. Uphoff C. C. et al., 2012. Treatment of mycoplasma contamination in cell cultures with Plasmocin. J Biomed Biotechnol. 2012:267678.
3. Rongvaux A. et al., 2014. Development and function of human innate immune cells in a humanized mouse model. Nat Biotechnol. 32(4):364-72.
4. Baronti C. et al., 2013. Mycoplasma removal: simple curative methods for viral supernatants. J Virol Methods. 187(2):234-7.
5. Deng F. et al. 2012. Generation of induced pluripotent stem cells from human Tenon's capsule fibroblasts. Mol Vis. 18:2871-81.
6. Romorini L. et al, 2013. Effect of antibiotics against Mycoplasma sp. on human embryonic stem cells undifferentiated status, pluripotency, cell viability and growth. PLoS One. 8(7):e70267/
Plasmocin® Prophylactic (2.5 mg/ml): to prevent mycoplasma contamination, use at 2.5 - 5 µg/ml on a regular basis.
Plasmocin® Treatment (25 mg/ml): to eliminate mycoplasmas, use at 25 µg/ml for 2 weeks.
Endotoxin level: < 2 EU/mg
Physicochemical characterization: pH and appearance
Cell culture tested: potency validated on bacterial reference strains
Plasmocin® is provided as a cell culture tested, sterile filtered yellow solution.
This product is available at different concentrations:
ant-mpp: 25 mg/ml (Plasmocin® Treatment)
ant-mpt: 2.5 mg/ml (Plasmocin® Prophylactic)
 Plasmocin® Prophylactic and Plasmocin® Treatment are shipped at room temperature
Plasmocin® Prophylactic and Plasmocin® Treatment are shipped at room temperature
 Upon receipt, the product can be stored at 4°C for 1 month or at -20°C for long-term storage.
Upon receipt, the product can be stored at 4°C for 1 month or at -20°C for long-term storage.
如需购买或咨询该产品技术问题,请联系Invivogen一级代理商-欣博盛生物